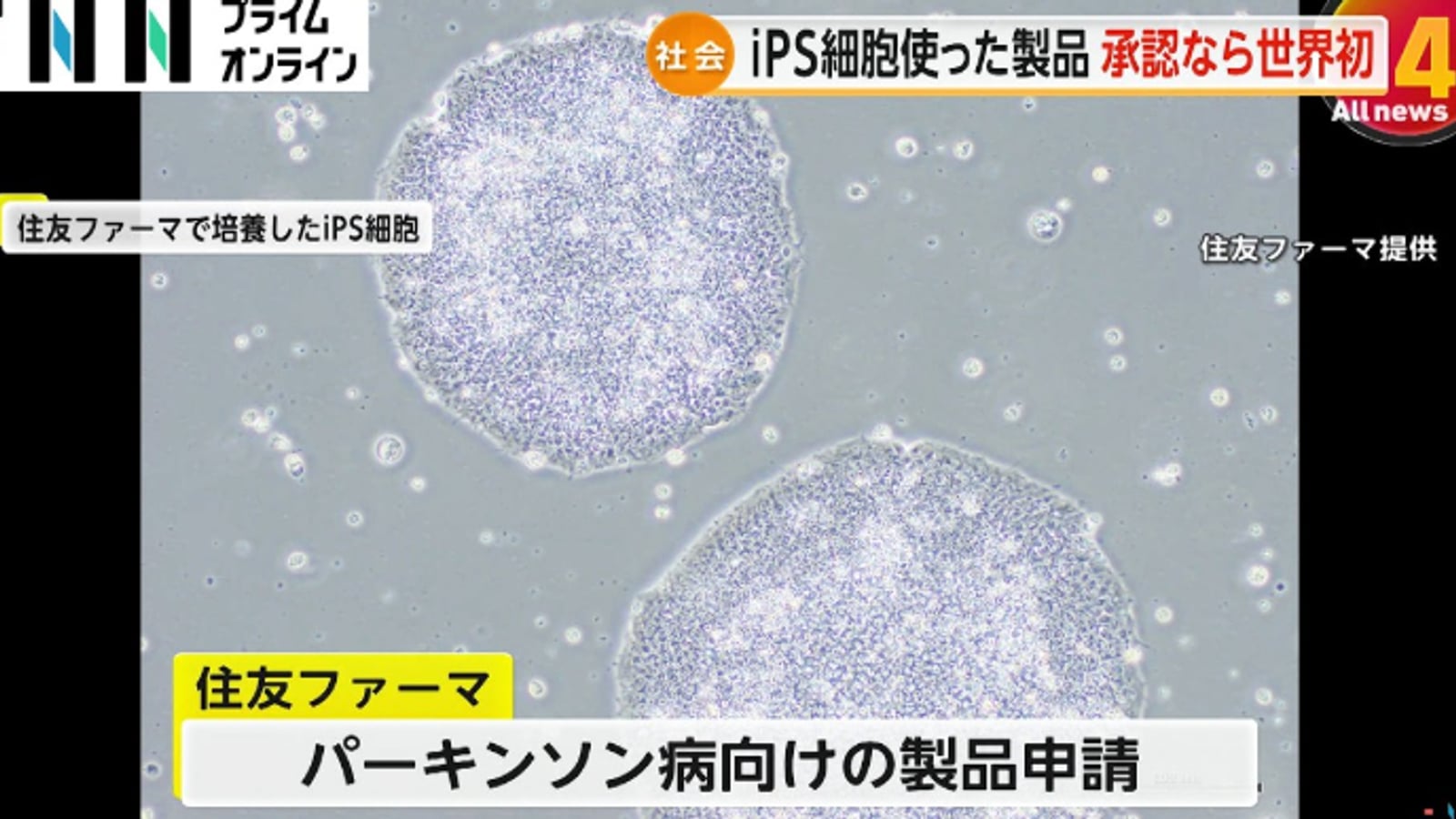
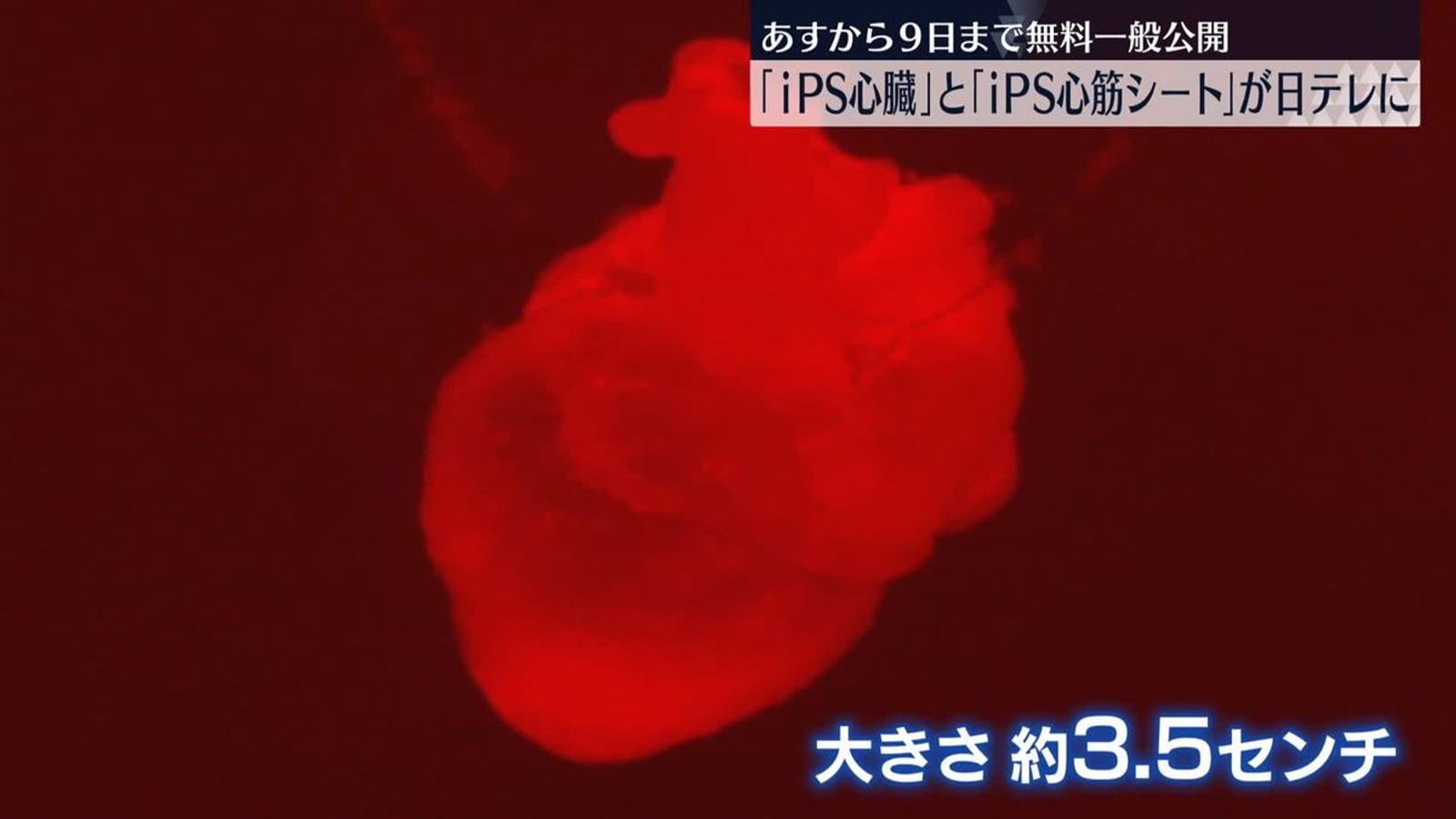
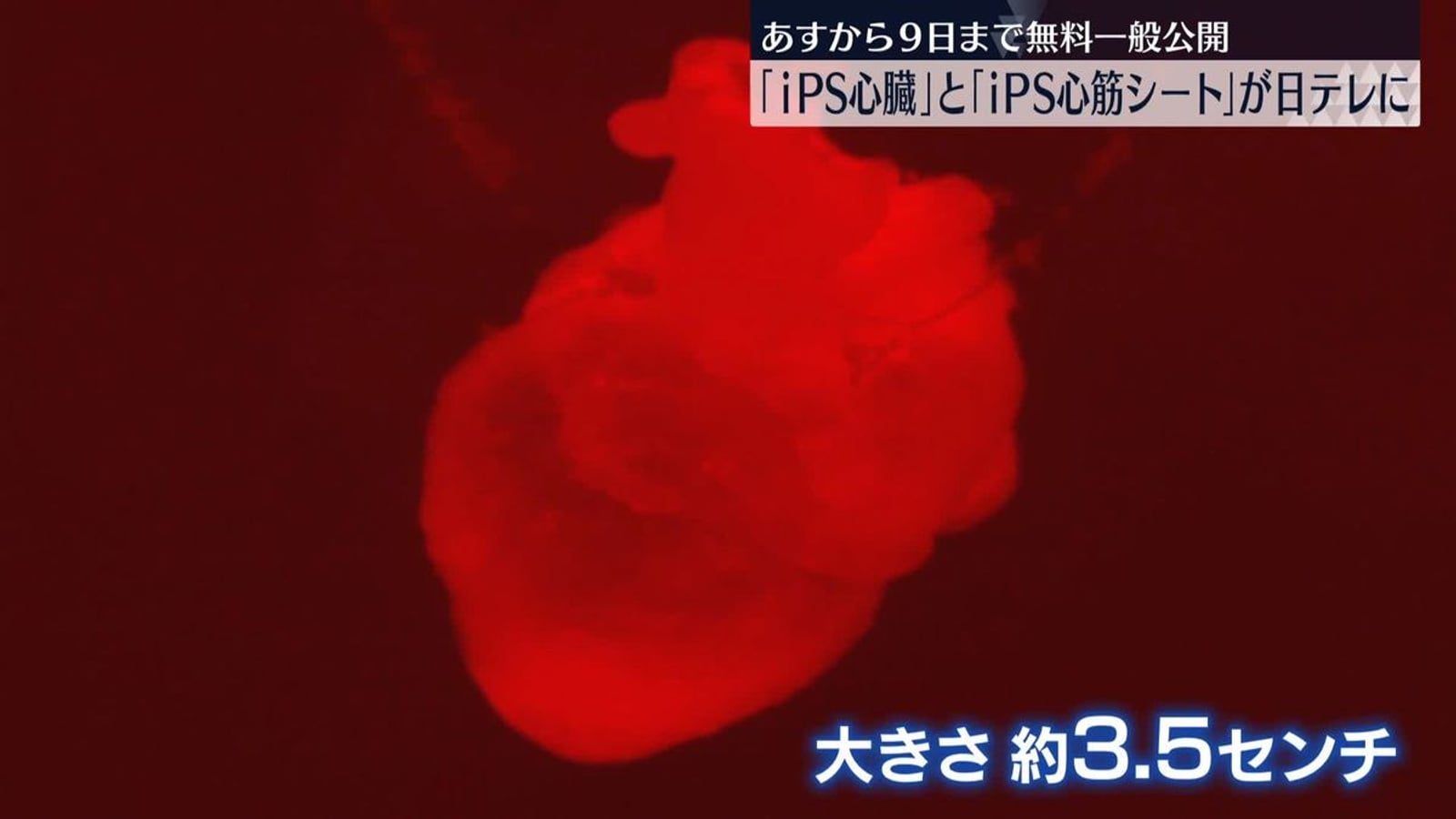
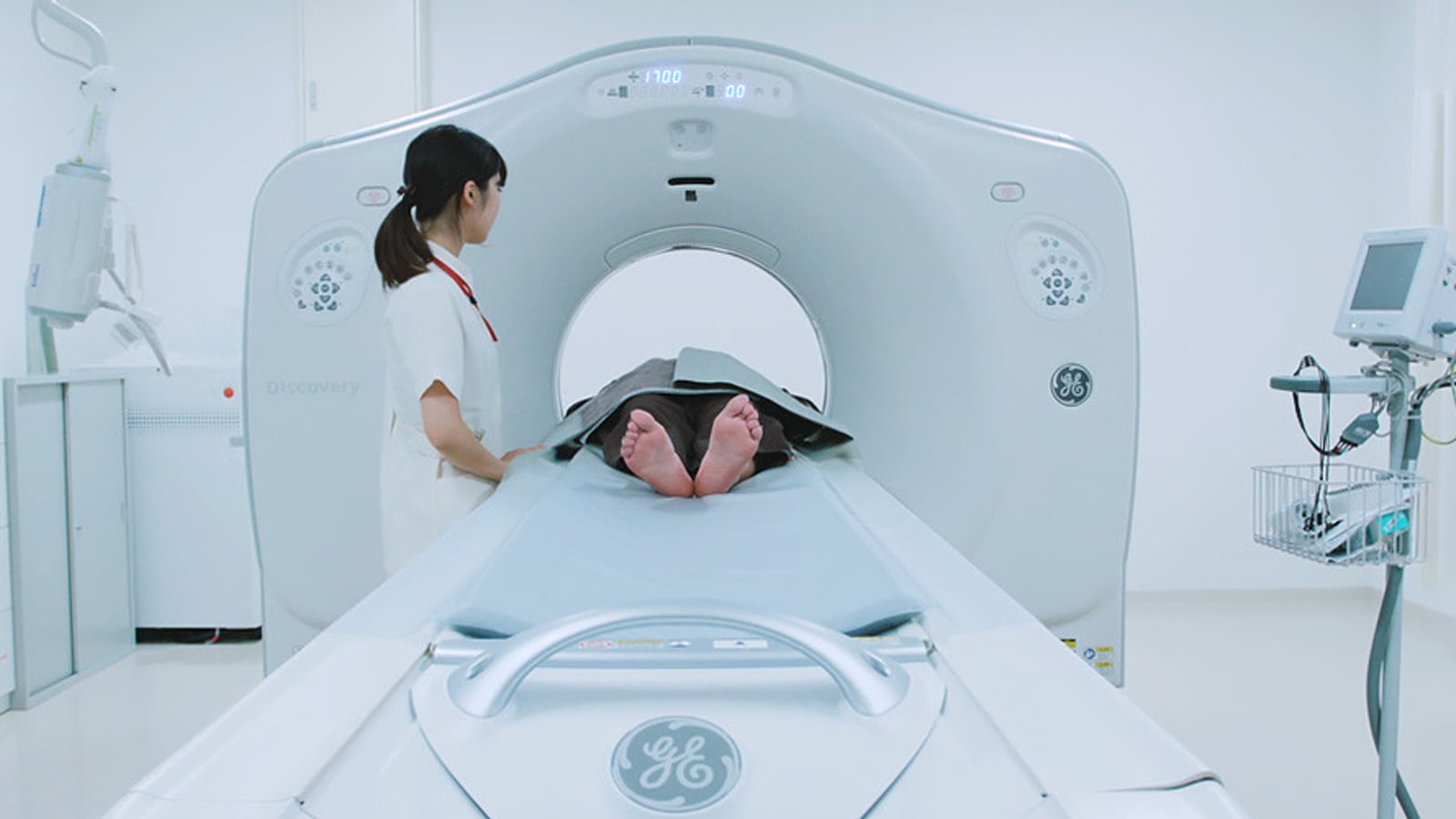
Visiting GINZA YR CLINIC in Tokyo Ginza: From Physician Background to Safety Compliance
From the Ginza location and clinic interior, to Dr. Junya Higuchi's background as a University of Tokyo Medical Doctor, compliance practices, and an overview of stem cell, PRP, immune cell, and NAD+ treatment directions.
Read More